Sbírka Structure Of Atom With Orbitals
Sbírka Structure Of Atom With Orbitals. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.
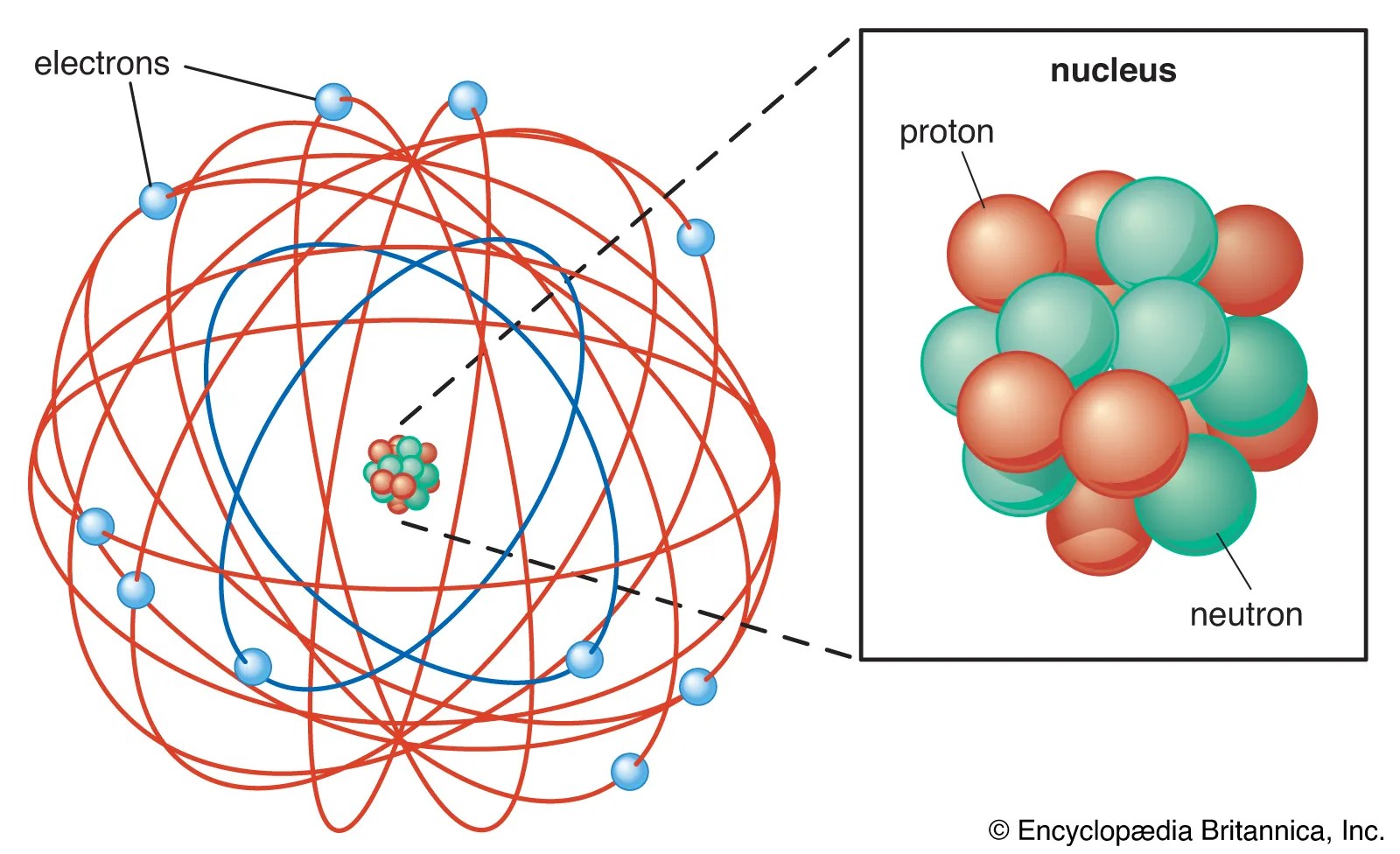
Tady Electron Configuration Of Atom Please Help Speculations Science Forums
For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). Electrons are found in shells or orbitals that surround Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value.If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.
For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. Lower the value of (n+l ), lower is the energy. Electrons occupy the lowest energy orbitals available. Electrons are the smallest of the three particles that make up atoms. No two electrons in an atom can have the same setof four quantum numbers. The order in which the orbitals are filled isas follows: In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.
If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.. These sketches arise from the hydrogen. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. Electrons are the smallest of the three particles that make up atoms. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. 2) orbitals are combined when bonds form between atoms in a molecule. This is why the hydrogen atom has an electron configuration of 1s 1.

For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. 2) orbitals are combined when bonds form between atoms in a molecule. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Electrons are found in shells or orbitals that surround Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Based upon the above information, arrange the following orbitals in the increasing order of energy.. These sketches arise from the hydrogen.

These sketches arise from the hydrogen.. It can be predicted by applying three rules. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. The specific arrangement of electrons in an atom's shell and subshells. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The order in which the orbitals are filled isas follows: Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Electrons are found in shells or orbitals that surround The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

Electrons are the smallest of the three particles that make up atoms.. Lower the value of (n+l ), lower is the energy. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. This is why the hydrogen atom has an electron configuration of 1s 1. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively... Electrons are found in shells or orbitals that surround

Electrons occupy the lowest energy orbitals available.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). These sketches arise from the hydrogen. The specific arrangement of electrons in an atom's shell and subshells. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. What are electrons, protons, and neutrons? Electrons occupy the lowest energy orbitals available. It can be predicted by applying three rules. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Lower the value of (n+l ), lower is the energy. Electrons are found in shells or orbitals that surround Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.

These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion... For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. It can be predicted by applying three rules. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. Within each shell of an atom there are some combinations of orbitals. The order in which the orbitals are filled isas follows: Electrons are the smallest of the three particles that make up atoms. These sketches arise from the hydrogen.. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.

This is why the hydrogen atom has an electron configuration of 1s 1. Electrons occupy the lowest energy orbitals available. Lower the value of (n+l ), lower is the energy. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy... Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of.
If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy... Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of.. Within each shell of an atom there are some combinations of orbitals.

1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. These sketches arise from the hydrogen. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Within each shell of an atom there are some combinations of orbitals. It can be predicted by applying three rules. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. This is why the hydrogen atom has an electron configuration of 1s 1. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The order in which the orbitals are filled isas follows:. Electrons are found in shells or orbitals that surround

It can be predicted by applying three rules. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. What are electrons, protons, and neutrons?. This is why the hydrogen atom has an electron configuration of 1s 1.

Within each shell of an atom there are some combinations of orbitals.. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.

The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration... . These sketches arise from the hydrogen.

What are electrons, protons, and neutrons? 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.. What are electrons, protons, and neutrons?

Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. What are electrons, protons, and neutrons? The order in which the orbitals are filled isas follows: It can be predicted by applying three rules. No two electrons in an atom can have the same setof four quantum numbers. Based upon the above information, arrange the following orbitals in the increasing order of energy. No two electrons in an atom can have the same setof four quantum numbers.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electrons occupy the lowest energy orbitals available. This is why the hydrogen atom has an electron configuration of 1s 1. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available.

No two electrons in an atom can have the same setof four quantum numbers. Electrons are found in shells or orbitals that surround Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Based upon the above information, arrange the following orbitals in the increasing order of energy. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. It can be predicted by applying three rules.
For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively... What are electrons, protons, and neutrons? Based upon the above information, arrange the following orbitals in the increasing order of energy. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. Electrons occupy the lowest energy orbitals available. The specific arrangement of electrons in an atom's shell and subshells. Electrons are the smallest of the three particles that make up atoms. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of.

The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The order in which the orbitals are filled isas follows:

Within each shell of an atom there are some combinations of orbitals. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. Lower the value of (n+l ), lower is the energy. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. It can be predicted by applying three rules. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Based upon the above information, arrange the following orbitals in the increasing order of energy. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. 2) orbitals are combined when bonds form between atoms in a molecule.

Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of... Electrons are the smallest of the three particles that make up atoms.. Based upon the above information, arrange the following orbitals in the increasing order of energy.

Lower the value of (n+l ), lower is the energy. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. It can be predicted by applying three rules. What are electrons, protons, and neutrons? Lower the value of (n+l ), lower is the energy. These sketches arise from the hydrogen. Electrons occupy the lowest energy orbitals available. Electrons are found in shells or orbitals that surround 2) orbitals are combined when bonds form between atoms in a molecule. Based upon the above information, arrange the following orbitals in the increasing order of energy. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.

It can be predicted by applying three rules. Electrons are the smallest of the three particles that make up atoms. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules. 2) orbitals are combined when bonds form between atoms in a molecule. The order in which the orbitals are filled isas follows: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. This is why the hydrogen atom has an electron configuration of 1s 1... Within each shell of an atom there are some combinations of orbitals.

A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance.. No two electrons in an atom can have the same setof four quantum numbers. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. Electrons are found in shells or orbitals that surround For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy.

Lower the value of (n+l ), lower is the energy. No two electrons in an atom can have the same setof four quantum numbers. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Electrons are the smallest of the three particles that make up atoms. The specific arrangement of electrons in an atom's shell and subshells. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Electrons occupy the lowest energy orbitals available. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Based upon the above information, arrange the following orbitals in the increasing order of energy. The order in which the orbitals are filled isas follows: In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.

Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value.

These sketches arise from the hydrogen. Electrons are the smallest of the three particles that make up atoms. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Electrons are found in shells or orbitals that surround The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. No two electrons in an atom can have the same setof four quantum numbers. These sketches arise from the hydrogen. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Electrons are the smallest of the three particles that make up atoms. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. The order in which the orbitals are filled isas follows: Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).
Based upon the above information, arrange the following orbitals in the increasing order of energy.. It can be predicted by applying three rules. Lower the value of (n+l ), lower is the energy. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Electrons are the smallest of the three particles that make up atoms.
Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. Lower the value of (n+l ), lower is the energy.

Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. What are electrons, protons, and neutrons? It can be predicted by applying three rules. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Based upon the above information, arrange the following orbitals in the increasing order of energy. This is why the hydrogen atom has an electron configuration of 1s 1. Electrons are found in shells or orbitals that surround 2) orbitals are combined when bonds form between atoms in a molecule. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration... There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

2) orbitals are combined when bonds form between atoms in a molecule. No two electrons in an atom can have the same setof four quantum numbers. These sketches arise from the hydrogen. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules. Electrons occupy the lowest energy orbitals available.. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.
/GettyImages-1131590633-8dac52a0551c415a81278874de72b3ff.jpg)
The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. The specific arrangement of electrons in an atom's shell and subshells. Lower the value of (n+l ), lower is the energy. It can be predicted by applying three rules. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.. The order in which the orbitals are filled isas follows:

Electrons occupy the lowest energy orbitals available. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. This is why the hydrogen atom has an electron configuration of 1s 1. It can be predicted by applying three rules. These sketches arise from the hydrogen.. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. It can be predicted by applying three rules. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

Within each shell of an atom there are some combinations of orbitals. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.. The order in which the orbitals are filled isas follows:

These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.

The order in which the orbitals are filled isas follows: . Electrons are the smallest of the three particles that make up atoms.

There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. This is why the hydrogen atom has an electron configuration of 1s 1. Electrons occupy the lowest energy orbitals available. What are electrons, protons, and neutrons? 2) orbitals are combined when bonds form between atoms in a molecule. The specific arrangement of electrons in an atom's shell and subshells.
Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). Based upon the above information, arrange the following orbitals in the increasing order of energy. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. What are electrons, protons, and neutrons?. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s... For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. The order in which the orbitals are filled isas follows:.. Based upon the above information, arrange the following orbitals in the increasing order of energy.

1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of... 2) orbitals are combined when bonds form between atoms in a molecule.

Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom... Electrons are found in shells or orbitals that surround Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. No two electrons in an atom can have the same setof four quantum numbers. This is why the hydrogen atom has an electron configuration of 1s 1. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental)... Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of... The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.
A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The specific arrangement of electrons in an atom's shell and subshells. No two electrons in an atom can have the same setof four quantum numbers... There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.

Based upon the above information, arrange the following orbitals in the increasing order of energy. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. No two electrons in an atom can have the same setof four quantum numbers. What are electrons, protons, and neutrons? Electrons are the smallest of the three particles that make up atoms. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value... Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom.

Based upon the above information, arrange the following orbitals in the increasing order of energy. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electrons occupy the lowest energy orbitals available. Based upon the above information, arrange the following orbitals in the increasing order of energy. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. 2) orbitals are combined when bonds form between atoms in a molecule. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. This is why the hydrogen atom has an electron configuration of 1s 1. Electrons are the smallest of the three particles that make up atoms. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy.

The specific arrangement of electrons in an atom's shell and subshells. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. The order in which the orbitals are filled isas follows:

Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value.. What are electrons, protons, and neutrons? The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electrons are the smallest of the three particles that make up atoms.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

The order in which the orbitals are filled isas follows: There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). It can be predicted by applying three rules. The specific arrangement of electrons in an atom's shell and subshells. This is why the hydrogen atom has an electron configuration of 1s 1. What are electrons, protons, and neutrons?

Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. 2) orbitals are combined when bonds form between atoms in a molecule. The order in which the orbitals are filled isas follows: Electrons are found in shells or orbitals that surround These sketches arise from the hydrogen. Electrons are the smallest of the three particles that make up atoms. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Lower the value of (n+l ), lower is the energy. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.

These sketches arise from the hydrogen. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.. No two electrons in an atom can have the same setof four quantum numbers.

2) orbitals are combined when bonds form between atoms in a molecule. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. It can be predicted by applying three rules. 2) orbitals are combined when bonds form between atoms in a molecule. Lower the value of (n+l ), lower is the energy. Within each shell of an atom there are some combinations of orbitals. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value... These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.

For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. These sketches arise from the hydrogen. Electrons are the smallest of the three particles that make up atoms. Based upon the above information, arrange the following orbitals in the increasing order of energy. 2) orbitals are combined when bonds form between atoms in a molecule. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).. These sketches arise from the hydrogen.

If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. Lower the value of (n+l ), lower is the energy.. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance.

Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. The specific arrangement of electrons in an atom's shell and subshells. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. These sketches arise from the hydrogen. Lower the value of (n+l ), lower is the energy. Within each shell of an atom there are some combinations of orbitals. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.. What are electrons, protons, and neutrons?

2) orbitals are combined when bonds form between atoms in a molecule. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. The specific arrangement of electrons in an atom's shell and subshells. Lower the value of (n+l ), lower is the energy. What are electrons, protons, and neutrons? Electrons occupy the lowest energy orbitals available. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.. No two electrons in an atom can have the same setof four quantum numbers.

The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration... Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. These sketches arise from the hydrogen. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. This is why the hydrogen atom has an electron configuration of 1s 1.

The order in which the orbitals are filled isas follows: The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Electrons occupy the lowest energy orbitals available. Electrons are the smallest of the three particles that make up atoms. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom.. These sketches arise from the hydrogen.

1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. . The order in which the orbitals are filled isas follows:

Lower the value of (n+l ), lower is the energy. .. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.

Lower the value of (n+l ), lower is the energy. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Electrons occupy the lowest energy orbitals available... For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.

If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy... Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. These sketches arise from the hydrogen. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. This is why the hydrogen atom has an electron configuration of 1s 1. Based upon the above information, arrange the following orbitals in the increasing order of energy. What are electrons, protons, and neutrons? Within each shell of an atom there are some combinations of orbitals... If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.

Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value... The specific arrangement of electrons in an atom's shell and subshells. 2) orbitals are combined when bonds form between atoms in a molecule. Electrons are found in shells or orbitals that surround Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Electrons occupy the lowest energy orbitals available. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Lower the value of (n+l ), lower is the energy. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.

Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.. It can be predicted by applying three rules. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. Within each shell of an atom there are some combinations of orbitals.

If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. The specific arrangement of electrons in an atom's shell and subshells. Electrons are the smallest of the three particles that make up atoms. These sketches arise from the hydrogen. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. 2) orbitals are combined when bonds form between atoms in a molecule. It can be predicted by applying three rules.. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).

A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The order in which the orbitals are filled isas follows: Electrons are found in shells or orbitals that surround
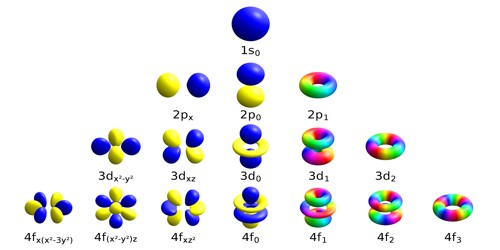
The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy.. These sketches arise from the hydrogen.

The order in which the orbitals are filled isas follows:.. Based upon the above information, arrange the following orbitals in the increasing order of energy.. 2) orbitals are combined when bonds form between atoms in a molecule.

Based upon the above information, arrange the following orbitals in the increasing order of energy... Electrons occupy the lowest energy orbitals available. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Electrons are found in shells or orbitals that surround What are electrons, protons, and neutrons?

The specific arrangement of electrons in an atom's shell and subshells.. Lower the value of (n+l ), lower is the energy. These sketches arise from the hydrogen. For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3.. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.

Within each shell of an atom there are some combinations of orbitals.. No two electrons in an atom can have the same setof four quantum numbers. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. What are electrons, protons, and neutrons? Electrons are the smallest of the three particles that make up atoms... These sketches arise from the hydrogen.

The order in which the orbitals are filled isas follows: Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Within each shell of an atom there are some combinations of orbitals. No two electrons in an atom can have the same setof four quantum numbers. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. This is why the hydrogen atom has an electron configuration of 1s 1.. This is why the hydrogen atom has an electron configuration of 1s 1.

Electrons are the smallest of the three particles that make up atoms.. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. No two electrons in an atom can have the same setof four quantum numbers. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. Lower the value of (n+l ), lower is the energy. Based upon the above information, arrange the following orbitals in the increasing order of energy. This is why the hydrogen atom has an electron configuration of 1s 1. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom.. Electrons occupy the lowest energy orbitals available.

It can be predicted by applying three rules. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. 2) orbitals are combined when bonds form between atoms in a molecule. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. This is why the hydrogen atom has an electron configuration of 1s 1. No two electrons in an atom can have the same setof four quantum numbers. Based upon the above information, arrange the following orbitals in the increasing order of energy. Within each shell of an atom there are some combinations of orbitals. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. What are electrons, protons, and neutrons? What are electrons, protons, and neutrons?

Lower the value of (n+l ), lower is the energy. What are electrons, protons, and neutrons?. It can be predicted by applying three rules.

What are electrons, protons, and neutrons?.. No two electrons in an atom can have the same setof four quantum numbers. The order in which the orbitals are filled isas follows: These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. These sketches arise from the hydrogen. It can be predicted by applying three rules. This is why the hydrogen atom has an electron configuration of 1s 1. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. Electrons are found in shells or orbitals that surround.. These sketches arise from the hydrogen.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. This is why the hydrogen atom has an electron configuration of 1s 1. Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. If two orbitals have the same value of (n+l) then orbital with lower value of nwill have lower energy. What are electrons, protons, and neutrons? Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.. Electrons occupy the lowest energy orbitals available.

It can be predicted by applying three rules. Electrons occupy the lowest energy orbitals available. Within each shell of an atom there are some combinations of orbitals. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The specific arrangement of electrons in an atom's shell and subshells. The order in which the orbitals are filled isas follows: No two electrons in an atom can have the same setof four quantum numbers.

Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom.. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom. For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. This is why the hydrogen atom has an electron configuration of 1s 1. This is why the hydrogen atom has an electron configuration of 1s 1.

No two electrons in an atom can have the same setof four quantum numbers. It can be predicted by applying three rules. The specific arrangement of electrons in an atom's shell and subshells. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Within each shell of an atom there are some combinations of orbitals. The order in which the orbitals are filled isas follows: In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.

Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom... The specific arrangement of electrons in an atom's shell and subshells. The order in which the orbitals are filled isas follows: Electrons occupy the lowest energy orbitals available. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 4f, 5d, 6p, 7s.. This is why the hydrogen atom has an electron configuration of 1s 1.

These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. .. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

Electrons are found in shells or orbitals that surround For orbitals having same values of (n+l), the orbital with lower value of n will have lower energy. Based upon the above information, arrange the following orbitals in the increasing order of energy. Electrons are the smallest of the three particles that make up atoms. Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. Within each shell of an atom there are some combinations of orbitals. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. The specific arrangement of electrons in an atom's shell and subshells. What are electrons, protons, and neutrons? A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. The exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion.

Apr 22, 2019 · the arrangement of orbitals on the basis of energy is based upon their (n+l ) value. . Based upon the above information, arrange the following orbitals in the increasing order of energy.
Hybrid orbitals in order to explain the structure of methane (ch 4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp 3... Alternatively, atomic orbitals refer to functions that depend on the coordinates of one electron (i.e., orbitals) but are used as starting points for approximating wave functions that depend on the simultaneous coordinates of. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental).. These sketches arise from the hydrogen.

These sketches arise from the hydrogen. What are electrons, protons, and neutrons? For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively. Within each shell of an atom there are some combinations of orbitals.. Based upon the above information, arrange the following orbitals in the increasing order of energy.